New NIR models speedup feedstock analysis process

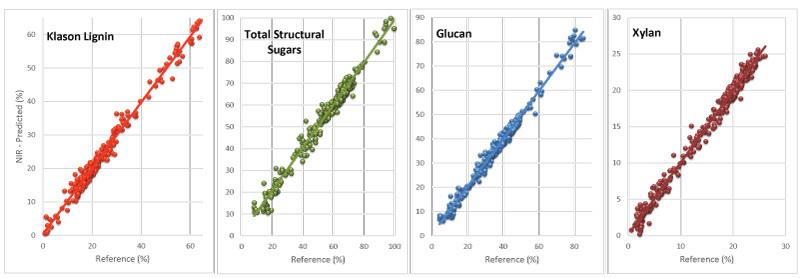

PHOTO: CELIGNIS
September 15, 2015
BY Daniel Hayes
Advertisement
Advertisement
Related Stories
Indigo Ag and Red Trail Energy have announced a collaboration to source low carbon intensity (CI) corn to support farmers using sustainable practices and benefit from emerging clean fuels market tax credit programs.
A record volume of sugarcane was processed during Brazil’s recently completed 2023-’24 harvest season, according to UNICA, the Brazilian sugarcane industry association. Ethanol production also set a new record, with a significant boost from corn.
The Renewable Fuels Association on April 15 urged the USDA’s National Agricultural Statistics Service to reconsider its plan to stop reporting farm acreage, yield and production estimates at the county level.
The USDA increased its forecast for 2023-’24 corn use in ethanol production in its latest World Agricultural Supply and Demand Estimates report, released April 11. The agency also lowered its forecast for season-average corn prices.
Farmers Edge Inc. and Gevo Inc. will work with growers spanning three states to track and quantify the carbon intensity impact of climate-smart practices helping to accelerate the production of SAF and low-CI ethanol,



