Searching for effective, environmentally friendly water treatment Technologies

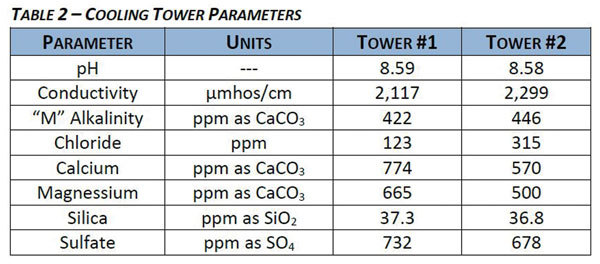
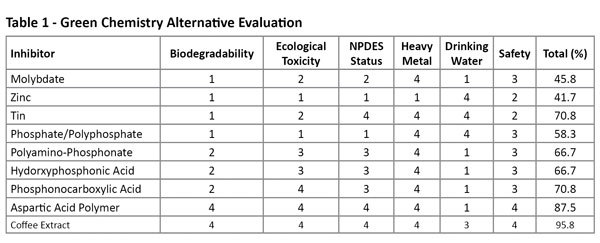


PHOTO: U.S. WATER SERVICES
September 17, 2015
BY Michael Mowbray
Advertisement
Advertisement
Related Stories
Valero Energy Corp. on April 25 announced that its SAF project in Texas is progressing ahead of schedule and expected to be operational this year. The company also reported its ethanol and renewable diesel operations were profitable during Q1.
U.S. fuel ethanol production fell by 3% the week ending April 19, according to data released by the U.S. Energy Information Administration on April 24. Stocks of fuel ethanol were down 1% and exports fell by 23%.
U.S. fuel ethanol production was down 7% the week ending April 12, according to data released by the U.S. Energy Information Administration on April 17. Stocks of fuel ethanol were down slightly and exports expanded by more than 12%.
A record volume of sugarcane was processed during Brazil’s recently completed 2023-’24 harvest season, according to UNICA, the Brazilian sugarcane industry association. Ethanol production also set a new record, with a significant boost from corn.
The ethanol and ethanol co-product industry continues to be a main economic driver in Nebraska, producing near-historic averages in 2020, despite lower ethanol prices and COVID-related production issues, according to a new study.





